|
|

|

|
|
|
 |
Membrane Protein Solutions |
|
|
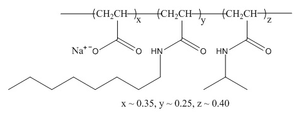
Amphipols & Detergents |
|
|
||||
|
|
||||
|
||||
|
|
||||
|
||||
|
Its 96 detergents were thoroughly selected from published crystallization trials in which they were shown to efficiently solubilize the membrane protein without shielding surface regions that are involved in crystal contacts. Discover JBScreen Detergents and receive 20 % discount on JBScreen Detergents HTS (Cat. No. CS-525) !* * Valid until May 12th, 2017, please indicate the code "Detergents promo" when placing your order!
|
||||
|
|
||||
| Responsible for content / Imprint | ||||
 Jena Bioscience GmbH
Jena Bioscience GmbH Loebstedter Str. 71 07749 Jena Germany Phone: +49 – 3641 – 6285 000 Fax: +49 – 3641 – 6285 100 E-Mail: info@jenabioscience.com Register Court: Amtsgericht Jena, HRB 207171 VAT No.: DE 195825742 Managing Directors: Thomas Billert Dr. Mathias Grün |
||||
| www.jenabioscience.com | ||||